|
Trend-wise, as one moves from left to right across a period in the modern periodic table, the electronegativity increases as the nuclear charge increases and the atomic size decreases. According to this scale, fluorine is the most electronegative element, while cesium is the least electronegative element. The scale has been named the Pauling scale in his honour. The most commonly used scale to measure electronegativity was designed by Linus Pauling. It is a dimensionless property because it is only a tendency. The tendency of an atom in a molecule to attract the shared pair of electrons towards itself is known as electronegativity. Main article: Electronegativity Periodic variation of Pauling electronegativities In that case, the ionization energy decreases as atomic size increases due to adding a valence shell, thereby diminishing the nucleus's attraction to electrons. However, suppose one moves down in a group. The decrease in the atomic size results in a more potent force of attraction between the electrons and the nucleus. Trend-wise, as one moves from left to right across a period in the modern periodic table, the ionization energy increases as the nuclear charge increases and the atomic size decreases. The energy needed to remove the second electron from the neutral atom is called the second ionization energy and so on. The first ionization energy is the amount of energy that is required to remove the first electron from a neutral atom. It is also referred to as ionization potential. The ionization energy is the minimum amount of energy that an electron in a gaseous atom or ion has to absorb to come out of the influence of attracting force of the nucleus. These trends give a qualitative assessment of the properties of each element. These trends exist because of the similar electron configurations of the elements within their respective groups or periods they reflect the periodic nature of the elements. Major periodic trends include atomic radius, ionization energy, electron affinity, electronegativity, valency and metallic character. They were discovered by the Russian chemist Dmitri Mendeleev in the year 1863. In chemistry, periodic trends are specific patterns that are present in the periodic table that illustrate different aspects of certain elements when grouped by period and/or group. Point to Remember: Mostly for transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core.Specific recurring patterns that are present in the modern periodic table The periodic trends in properties of elements. (See the above or below periodic table to see the valence electrons of inner transition metals). Inner transition elements can have valence electrons ranging from 3 to 16. In some inner transition metals, the electrons of incomplete d-orbitals are also considered as valence electrons. Hence, for inner transition elements, the electrons of both f-subshells as well as s-subshell behave like valence electrons. The inner transition elements have incomplete f- subshells and they are very close to the outer s-subshell. 
(See the above or below periodic table to see the valence electrons of transition metals).Īlso the two bottom rows at the bottom of the periodic table are the inner transition elements (or f-block elements) also have the similar case. Hence, the transition elements (i.e d-block elements from group 3 to 12) can have more valence electrons ranging from 3 to 12. So, the electrons of both d-subshell and s-subshell behave like valence electrons. 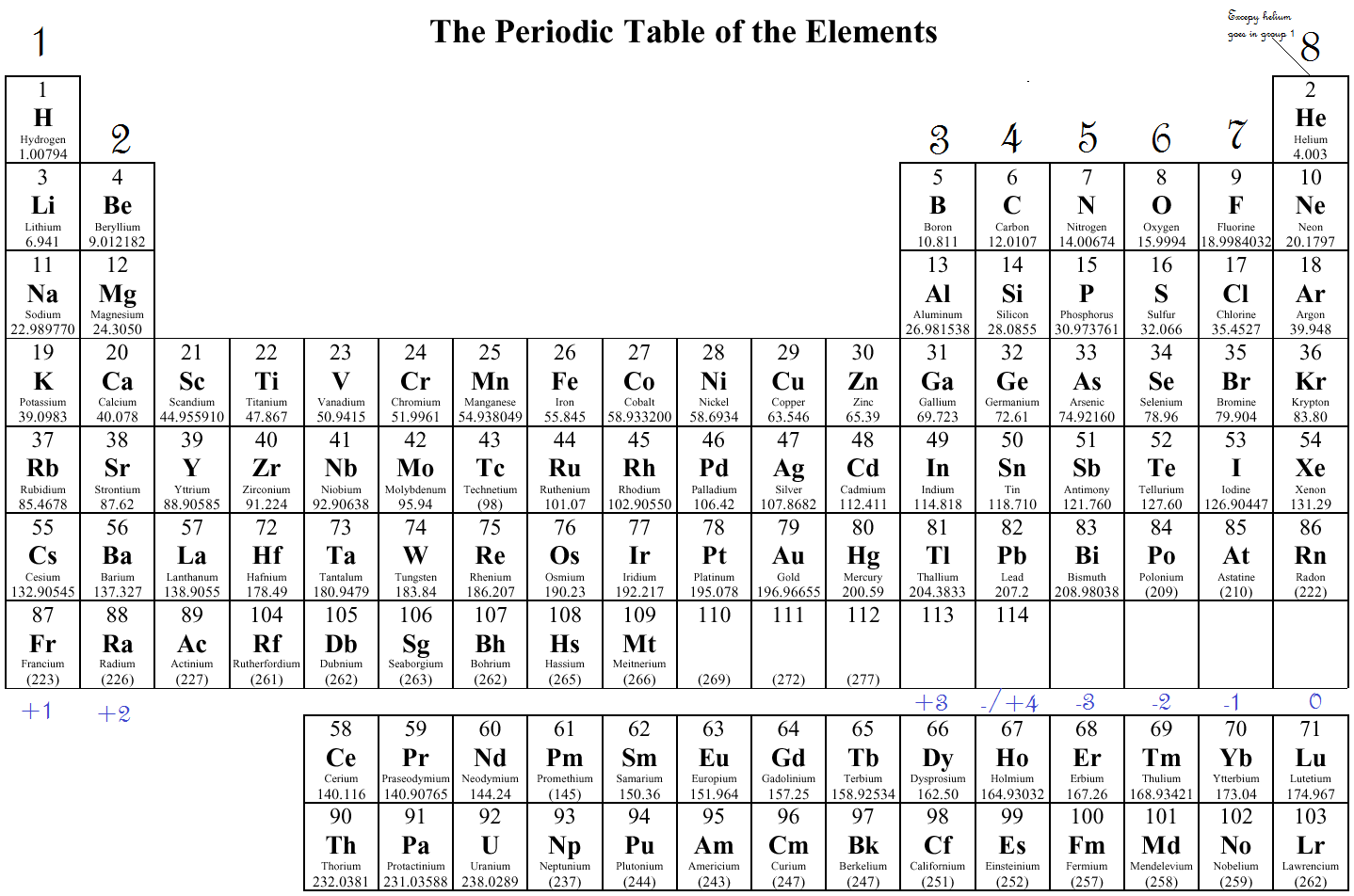
It is more difficult to find the valence electrons of transition elements as they have incompletely filled d-subshell and this d-subshell is very close to the outer s-subshell. What about valence electrons of transition and inner transition elements?įor the transition elements and inner transition elements, the case is more complicated. Well, this suits perfectly for the main group elements (i.e group 1, 2 and group 13 to 18), but what about the transition and inner transition elements? Hence, magnesium has 2 valence electrons. Here, you can see that the highest principal quantum number is 3, and the total electrons in this principal quantum number is 2. The electron configuration of magnesium is 1s 2 2s 2 2p 6 3s 2. Valence electrons can also be determined as the electrons present in the shell with highest principal quantum number (n). The magnesium element has 2 electrons in outermost orbit.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
